|
12/29/2023 0 Comments Group 7 reactivity trend 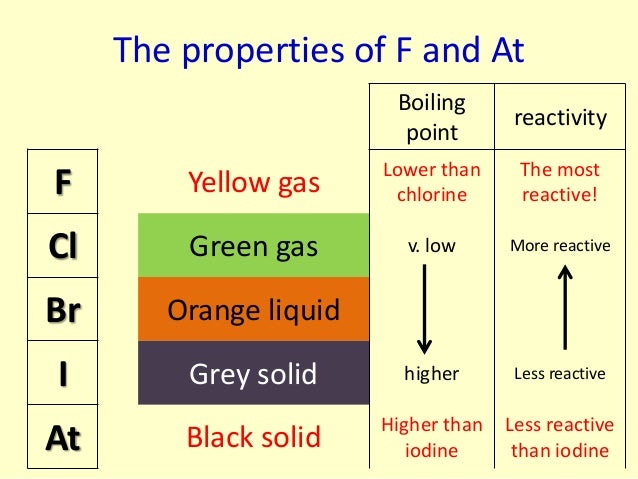
ĭiscusses the acidity of the hydrogen halides (like hydrogen chloride), and explains why HF is a weak acid.ĭescribes and explains the trend in reducing ability of the halide ions based on their reactions with concentrated sulphuric acid.ĭescribes and explains the tests for halide ions using silver nitrate solution followed by ammonia solution.ĭescribes the manufacture of chlorine by the electrolysis of sodium chloride solution using a diaphragm cell and a membrane cell. The reactions of halogens with hydrogen, phosphorus, sodium, iron, iron(II) ions, and sodium hydroxide solution. It also looks at the bond strengths of halogen-halogen bonds and of hydrogen-halogen bonds.ĭescribes and explains the trend in oxidising ability of the Group 7 elements based on the reactions between one halogen and the ions of another one - for example, between Cl 2 and I - ions from salts like KI. My reasons for not using the IUPAC system are discussed on this page in the Questions and Comments section.ĭiscusses trends in atomic radius, electronegativity, electron affinity and melting and boiling points of the Group 7 elements. Note: If you use the current IUPAC (international Union of Pure and Applied Chemistry) system for group numbering, you will probably know what I call Group 7 as Group 17. Core Chemistry 14 - 16 The Periodic Table - the Halogens This page introduces the Halogens in Group 7 of the Periodic Table. Periodic Table Group 7 menu Understanding Chemistry (b) Describe what you would observe when aqueous silver nitrate is added to separate aqueous solutions of Potassium Fluoride and Potassium Bromide. 
(ii) Explain why Bromine does not react with aqueous Chloride ions. Write an equation for the reaction occurring. (a) (i) Describe what you would observe when an aqueous solution of Bromine is added to an aqueous solution containing Iodide ions. As with group 1 and 2, the trends in properties and. So there will be no reaction in the I 2/KCl mixture or the I 2/KBr mixtureīoth tubes takes the colour of the Iodine - brown if concentrated enough, yellow if very dilute. Iodine is the least reactive halogen (besides astatine which is often ignored because it is extremely rare). We should expect this because Iodine is larger than Br and Cl, so not capable of taking electrons from either ions. When KCl and KBr have Iodine water added there is no colour change in either tube. The tube takes the colour of the Iodine - brown if concentrated enough, yellow if very dilute The last three tubes: Adding Iodine water to KI is a waste of time. This means that in the Br 2/KI mixture Bromine will be converted to Bromide ions while and Iodine will be made from Iodide ions.īut there will be no reaction in the Br 2/KCl mixtureīr 2(aq) + 2I - (aq) → 2Br - (aq) + I 2(aq) These demonstrations show the similarity of the physical and chemical properties of the alkali metals and the trend in reactivity down group 1 of the Periodic Table. So, Bromine is capable of taking electrons from Br - ions but not from Cl - ions. We should expect this because Bromine is smaller than I but not Cl. When KCl and KI have Bromine water added there is a colour change in one tube only. The next three tubes Adding Bromine water to KBr is a waste of time. (note that Group 1 halide solutions have no colour ) The reactivity of Group 7 non-metals increases as you move up the group Out of the 3 halogens, chlorine, bromine and iodine, chlorine is the most reactive and iodine is the least reactive Observations in aqueous solution The colour of the solution in the test tube shows which free halogen is present in solution. Observations: Slightly green/yellow solution goes orange. Observations: Slightly green/yellow solution goes brownĬl 2(aq) + 2Br - (aq) → 2Cl - (aq) + Br 2(aq) This means that Chlorine will be converted to Chloride ions while Bromine and Iodine will be made from Bromide and Iodide ions.Ĭl 2(aq) + 2I - (aq) → 2Cl - (aq) + I 2(aq) 
We should expect this because Chlorine is smaller than Br or I, so capable of taking electrons from Br - or I - ions. The next two tubes: When KBr and KI are added to Chlorine water there is a colour change in both tubes. The tube stays colourless (or at least very pale yellow) The first tube: Adding Chlorine water to KCl is a waste of time. This is an experiment you may well do, or have already done. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
